Forecyte Bio Limited
US:
7495 New Horizon Way, Suite 130-150, Frederick,MD 21703
Tel.: +1 215-589-3593
China:
177 Yiwei Road, Pilot free trade zone, Shanghai, China
Tel.:+86 21 80438700
E-mail:info@forecytebio.com
Preclinical Grade
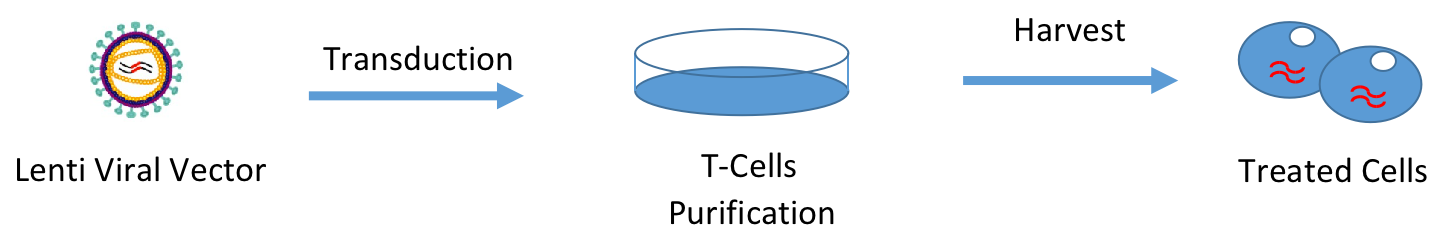
With our closed and automated system, Forecyte Bio offers high efficiency transduction process of genes to cells. Our experience enables us to handle several different kinds of cells such as CAR-T, TCR-T, CAR-NK.
GMP Grade
With our GMP facility in place, Forecyte is ready to provide production service under GMP conditions that meet the stringent requirements by different regulatory bodies such as FDA, NMPA, and EMA. Production scale supports clinical trials and commercial launch.
QC
Our quality is supported by our strict QA/QC policies and analytics; it includes
|
Category |
Attribute |
CAR-T |
General Cell |
|
Strength/Potency |
Cell concentration |
● |
● |
|
Cell viability |
● |
● |
|
|
Cytokine ELISA Titer |
● |
● |
|
|
CAR+% (Flow) |
● |
|
|
|
Identity |
Flow cytometry (cell CD marker typing) |
● |
● |
|
Purity |
Flow cytometry (cell CD marker typing %) |
● |
● |
|
Residual beads |
● |
● |
|
|
Residual vector (VSV-G) |
● |
● |
|
|
Safety |
Endotoxin |
● |
● |
|
Sterility (BACT/ALERT 3D) |
● |
● |
|
|
RCL (PCR) |
● |
● |
|
|
Mycoplasma (PCR) |
● |
● |
|
|
Residual tumor cell (Flow) |
● |
● |
|
|
General property |
Product appearance/color |
● |
● |
|
pH |
● |
● |
|
|
Osmolality |
● |
● |
Forecyte Bio Limited
US:
7495 New Horizon Way, Suite 130-150, Frederick,MD 21703
Tel.: +1 215-589-3593
China:
177 Yiwei Road, Pilot free trade zone, Shanghai, China
Tel.:+86 21 80438700
E-mail:info@forecytebio.com
Copyright © 2021 forecytebio.com All rights reserved.
沪ICP备2021022864号-1 power by:300.cn